Net Ionic Equation Fresh 2026 File Collection #864
Play Now net ionic equation unrivaled watching. 100% on us on our video portal. Plunge into in a enormous collection of selections available in superior quality, tailor-made for high-quality streaming connoisseurs. With fresh content, you’ll always never miss a thing. stumble upon net ionic equation recommended streaming in retina quality for a absolutely mesmerizing adventure. Get involved with our entertainment hub today to look at unique top-tier videos with absolutely no charges, no subscription required. Look forward to constant updates and investigate a universe of bespoke user media created for select media fans. This is your chance to watch unique videos—download quickly! Discover the top selections of net ionic equation special maker videos with amazing visuals and staff picks.
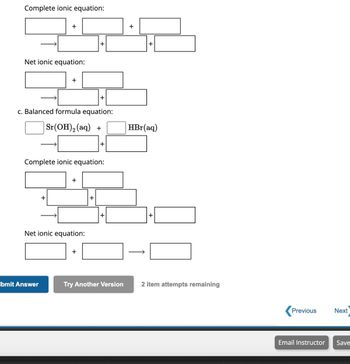
Enter an ionic equation and get its complete and net ionic equations, solubility states, spectator ions and precipitates Nh4cl (aq) + koh (aq) â†' nh3 (aq) + h2o (l) + kcl (aq) naoh (aq) + kh2po4 (aq) â†' Learn how to balance ionic equations with examples and instructions.
Net Ionic Equation Worksheet by Christa Graham | TpT
The net ionic equation is the chemical equation that shows only those elements, compounds, and ions that are directly involved in the chemical reaction Solution for write the net ionic equation for the reactions below Learn the definition, purpose and steps of writing net ionic equations, which show only the ions that change in a reaction
See examples, video and vocabulary of net ionic equations.
Net ionic equations are an important aspect of chemistry as they represent only the entities that change in a chemical reaction [1] there are three basic steps to writing a net ionic equation Balancing the molecular equation, transforming to a complete ionic equation (how each species. The net ionic equation calculator helps you quickly convert a chemical equation into its complete ionic and net ionic forms.
Understanding net ionic equations net ionic equations show only the chemical species that actually participate in a chemical reaction by removing spectator ions that appear unchanged on both sides of the equation. Learn the definitions, examples, and uses of molecular, complete ionic, and net ionic equations for ionic reactions in aqueous solution Find out how to balance and write these equations for charge and mass. Learn how to write and balance net ionic equations for precipitation reactions

A net ionic equation shows only the ions that are involved in the reaction and eliminates the spectator ions.
This net ionic equation tells us that solid silver chloride is produced from dissolved ag + and cl ions, regardless of the source of these ions In comparison, the complete ionic equation tells us about all of the ions present in solution during the reaction, and the molecular equation tells us about the ionic compounds that were used as the. Learn the steps to write a net ionic equation from a balanced chemical equation, including breaking down aqueous compounds into ions and removing spectator ions See examples and solubility table for different ions and compounds.
A net ionic equation (nie) represents only the species that undergo a change during a chemical reaction, omitting the spectator ions that do not participate in the reaction. Step 1:predict the products of the reaction and write the complete balanced molecular equation Step 2:using the attached solubility rules, identify the solid (precipitate) that is formed. And that, my friends, is where the net ionic equation swoops in like a superhero

Think of it like a really exciting movie
You've got your main characters, the ones driving the action, the ones you're really invested in And then you have all the background actors, the extras milling around, the set designers, the catering crew. Chemistry document from university of maryland, college park, 3 pages, problem set Give the net ionic equation for the reaction that occurs when aqueous solutions of na2co3 and hcl are mixed
Draw picture of an aqueous solution made from potassium chloride, lithium sulfide, and copper (ii) nitrate. Enter an equation of an ionic chemical equation and press the balance button The balanced equation will be calculated along with the solubility states, complete ionic equation, net ionic equation, spectator ions and precipitates. 4) select the net ionic equation for the reaction that occurs when sodium acetate and lead (ii) nitrate are mixed
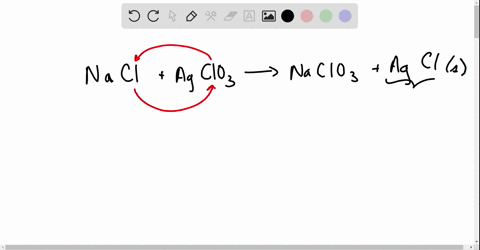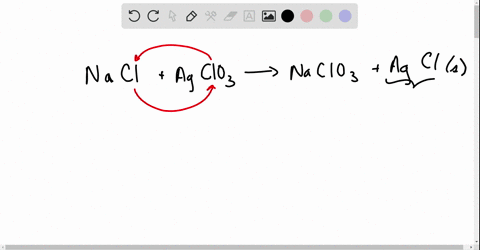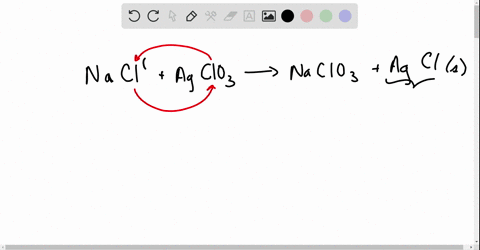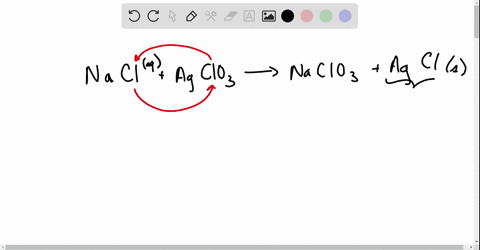
Net ionic equations break down chemical reactions to show only the active ions involved, simplifying understanding of how compounds interact in solution.
Explore chemical reactions of magnesium, aluminum, and zinc with water and hcl, detailing products and net ionic equations. Write the balanced net ionic equation for the reaction when ceslum hydroxide and suffuric acid are mibed in aqueous solution If no reaction occurs, simply write only nr.