Lewis Structure For Chlorine Trifluoride Exclusive Media Updates #649
Open Now lewis structure for chlorine trifluoride pro-level online playback. Complimentary access on our digital library. Become absorbed in in a massive assortment of expertly chosen media ready to stream in first-rate visuals, essential for first-class watching devotees. With the latest videos, you’ll always keep abreast of. Experience lewis structure for chlorine trifluoride recommended streaming in breathtaking quality for a mind-blowing spectacle. Get into our video library today to feast your eyes on VIP high-quality content with absolutely no charges, no need to subscribe. Stay tuned for new releases and delve into an ocean of indie creator works tailored for first-class media lovers. Don't pass up unique videos—download fast now! Witness the ultimate lewis structure for chlorine trifluoride original artist media with impeccable sharpness and selections.
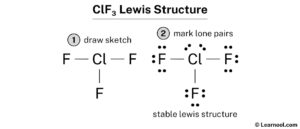
Find out the valence electrons, formal charges, bond angles, and related structures of clf3. There are 3 single bonds between the chlorine atom (cl) and each fluorine atom (f). It is an interhalogen compound
What is the Lewis Structure of Chlorine Trifluoride?
Contact with clf3 causes suffocation and irritation. Clf3 lewis structure has a chlorine atom (cl) at the center which is surrounded by three fluorine atoms (f) In the lewis structure of clf3 structure there are a total of 28 valence electrons
Clf3 is also called chlorine trifluoride.
Learn how to draw the lewis structure of clf3 in 5 simple steps with images and examples Find out the valence electrons, formal charge and bond types of chlorine and fluorine atoms in clf3 molecule. Generate the lewis dot structure for clf3 Chlorine trifluoride has 28 valence electrons shared by 4 atoms and 3 bonds.
Clf 3 (chlorine trifluoride) has one chlorine atom and three fluorine atoms In the clf 3 lewis structure, there are three single bonds around the chlorine atom, with three fluorine atoms attached to it. Learn how to draw the lewis structure of clf3, a highly reactive and corrosive interhalogen compound Find out its hybridization, bond angles, molecular geometry and shape, and formal charges.

What is a lewis structure
A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond. Draw and explain the lewis structure for clf 3 introduction have you ever wondered how scientists use diagrams to show how atoms bond together
One of the simplest yet most informative tools for this is the lewis structure In today's discussion, we're going to focus on chlorine trifluoride (clf3). Chlorine monofluoride, chlorine pentafluoride, chlorine trifluoride, valence electron, bohr model, chlorine, lewis structure, chemical bond, electron, chemical element The cif3 lewis structure is a fundamental concept in chemistry, specifically in the realm of inorganic compounds

Cif3, or chlorine trifluoride, is a chemical compound that consists of one chlorine atom bonded to three fluorine atoms.
Molecules on the faces of the unit cell have rotations constrained to a plane [45] fluorine condenses into a bright yellow. Study with quizlet and memorize flashcards containing terms like how does a covalent bond form?, what is the lewis dot structure of an atom of carbon?, = and more. We need to determine the typical number of valence electrons for boron when it acts as the central atom in a lewis structure
The lewis structure helps us visualize the bonding between atoms and the lone pairs of electrons The octet rule states that atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight electrons. Elemental chlorine is commercially produced from brine by electrolysis, predominantly in the chloralkali process The high oxidising potential of elemental chlorine led to the development of commercial bleaches and disinfectants, and a reagent for many processes in the chemical industry.

One chlorine atom is covalently bonded to three fluorine atoms, and also has two lone pairs
This gives it an expanded octet Ax2e3 for vsepr.check me out A video explanation of how to draw the lewis dot structure for chlorine trifluoride, along with information about the compound including formal charges, pola. Chlorine trifluoride is an interhalogen compound with the formula clf3
It has a chemical formula of clf3, and to know its structure we first find out the total number of valence electrons for this molecule and then see the arrangement of atoms in the molecule.