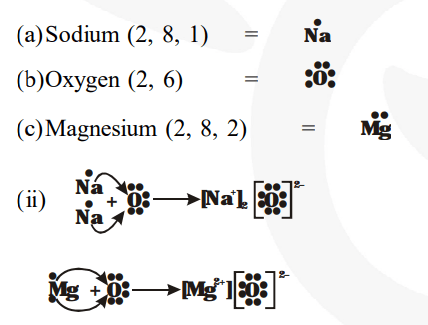Electron Dot Structure For Mg Last Update Content Files #608
Activate Now electron dot structure for mg deluxe on-demand viewing. Subscription-free on our digital collection. Plunge into in a comprehensive repository of documentaries available in top-notch resolution, the best choice for premium viewing connoisseurs. With hot new media, you’ll always have the latest info. Locate electron dot structure for mg organized streaming in vibrant resolution for a highly fascinating experience. Hop on board our streaming center today to check out restricted superior videos with zero payment required, free to access. Look forward to constant updates and venture into a collection of distinctive producer content intended for first-class media lovers. Grab your chance to see singular films—begin instant download! Access the best of electron dot structure for mg specialized creator content with dynamic picture and selections.
Generate the lewis dot structure for mg Al draw lewis structures for atoms of the following Enter a chemical element or formula to calculate and draw its lewis dot structure
Identify the correct Lewis electron-dot symbol for | Chegg.com
Be sure to use the proper capitalization for all element symbols Aluminum (al) aluminum is in group iiia (13) For the lewis structure of individual elements, use our valence electron calculator
The calculator will generate the lewis structure for known isomers along with the bonds, ionic charge, formal charge.
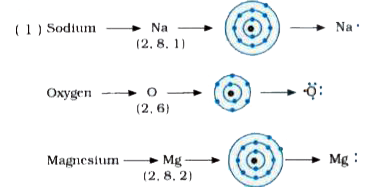
I show you where magnesium is on the periodic table and how to determine how many valence electrons magnesium has. Understanding electron dot diagrams for mg electron dot diagrams, also known as lewis diagrams or lewis dot structures, are a way to represent the valence electrons of an atom or ion These diagrams show the arrangement of electrons in the outermost energy level, also known as the valence shell. A lewis structure (also called lewis dot formula) is a diagram that shows the bonding between atoms and the lone pairs of electrons in a molecule
Bonds are shown as lines between atoms A single line for a single bond, double line for a double bond, and a triple line for a triple bond. There are two valence electrons in mg atom Hence, the lewis dot symbol for mg is
There is only one valence electron in an atom of sodium
Hence, the lewis dot structure is There are 3 valence electrons in boron atom There are six valence electrons in an atom of oxygen Learn how to draw the lewis dot diagram for magnesium and understand its electronic structure.
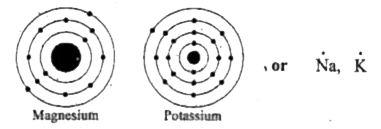
The electron dot structure of chlorine is given below When 2 chlorine atoms and magnesium atoms combine by transfer of electrons then magnesium chloride is formed by transfer of electrons Chlorine atom needs only one electron to complete its octet and magnesium cal removes two electrons to complete its octet. The lewis structure for mg (magnesium) shows two valence electrons surrounding the magnesium atom

This representation highlights magnesium's position as an alkaline earth metal, which readily loses these two electrons to form a mg²+ ion.
A lewis dot diagram, also known as an electron dot diagram or lewis structure, is a visual representation of the valence electrons in an atom or ion It is a simple way to illustrate the bonding and electron distribution in a chemical compound The lewis dot diagram for magnesium ion is a useful tool for understanding its chemical properties and reactivity Magnesium ion, represented by the.
It is a group two and period three element For determining its lewis structure, we first look at its valence electrons Find the lewis dot structure for magnesium in this video. Looking at lewis dot structure for ionic compounds worksheet answers isn't just about finding the right boxes to tick or the correct dots to draw

It's about understanding the fundamental forces that hold matter together.
(a) sulfur trioxide has more than one possible lewis (electron dot) structure (i) sketch a lewis (electron dot) structure for so 3 which obeys the octet rule [1] (ii) predict the length of each s to o bond in pm Use section 11 of the data booklet.
We need to write first the lewis dot structure.what should we do next?transfer the valence electrons of mg to s After the transfer of electron, what will happen to the elements? Nh3 lewis, epg vs mg valence The magnesium ion is represented without any dots, just its symbol and the positive charge.

Draw lewis structures for atoms of the following